Ex-pert Electrical Perspective
A Dynamic academic elaboration on the Art & Science of Hazardous Area Protection
Flash point & Auto-ignition Temperature
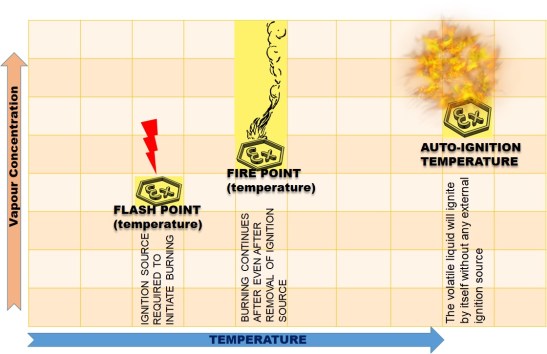
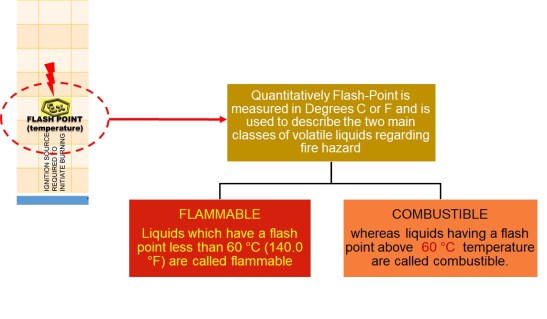
Flash Point
The Lowest temperature of a volatile ( Having tendency to change into vapors) liquid at which , under certain standard conditions, the liquid gives off vapors in a quantity such as to be capable of forming an ignitable vapor-air mixture.
(IEC 60079-10-1)
At the Flash Point Temperature, any ignition source influencing the vapors will cause the vapors to ignite and may cause an explosion.
Fire-Point
This the lowest temperature at which vapors of the material will keep burning after being ignited and the ignition source removed. The fire point is higher than the flash point, because at the flash point more vapor may not be produced rapidly enough to sustain combustion. (Wikipedia )
AUTO-IGNITION TEMPERATURE (Ignition temperature)
It is the lowest temperature of a mixture of flammable vapors with air , at which the mixture, due to its own high temperature , will ignite and explode without any external ignition process.
The mixture may attain high temperature (leading to auto-ignition ) due to following conditions (Few are listed as below):-
1. Contact with hot surface (For example : The globe or the protective glass of the light fixture, in operation)
2. High Ambient due to unexpected weather conditions and lack of ventilation.
The Auto-ignition Temperatures of some gases , much used in industry, are given below.
| Gas | Temperature |
|---|---|
| Methane | 580 °C |
| Hydrogen | 560 °C |
| Propane | 493 °C |
| Ethylene | 425 °C |
| Acetylene | 305 °C |
| Naphtha | 290 °C |
| Carbon disulphide | 102 °C |
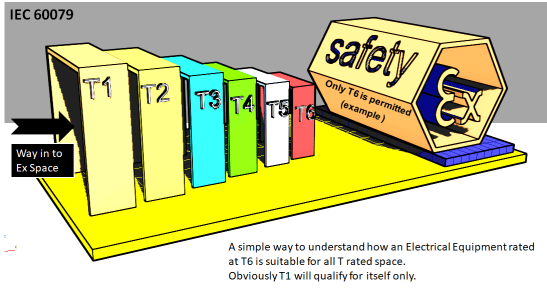
The temperatures as classified in the TEMPERATURE CLASS (IEC 60079-0) is actually the maximum surface temperature of the GROUP II equipment, which it may attain during operation but should not exceed the values as below:
| Temperature class | Maximum Surface Temperature ˚C |
| T 1 | 450 |
| T 2 | 300 |
| T 3 | 200 |
| T 4 | 135 |
| T 5 | 100 |
| T 6 | 85 |

The object is to compare the Auto-ignition temperature of the Gas , which is present in a particular area , with the Temperature Class of the Ex Certified equipment.
The Temperature Class of the equipment should be less than the Auto-Ignition Temperature of the mixture present in that area, so that during operation of the equipment the maximum temperature attained by the equipment remains below the auto-ignition temperature of the mixture.